|
2/19/2023 0 Comments Halogen periodic table Xenon: Xenon is a noble gas that has the symbol Xe. Halogen:Halogen refers to the chemical elements in the group 7 of the periodic table of elements. Both are included in the p block of the periodic table.ĭifference Between Halogen and Xenon Definition.This is because xenon can produce a beautiful blue light that is glowing when excited by electrical discharge. One of the major applications of xenon includes using as a light source. The appearance of xenon can be explained as a colorless and odorless gas. This reaction includes the reaction between xenon fluorides and water to produce xenon oxides and hydrogen fluoride (HF). But xenon cannot directly react with molecular oxygen. In addition, xenon also forms oxides by hydrolysis. 
Some of the fluorides that xenon can form are XeF 2, XeF 4 and XeF 6. For example, xenon can form halides by combining with halogens such as fluorine. One important characteristic of xenon is that it can form compounds under extreme conditions where most other noble gases cannot. It is very much stable and is less reactive. Therefore, the outermost orbital of xenon is completely filled with electrons. The electron configuration of xenon is given as 4d 105s 25p 6. It exists as a monoatomic gas in the standard temperature and pressure conditions. Xenon atom is a very large atom compared to other noble gases. It is included in the p block of the periodic table and is in the group 8. Xenon is a noble gas that has the symbol Xe. But other elements of halogen group can have up to +7 oxidation state. Fluorine can have only -1 and 0 oxidation states. Fluorine has a light greenish yellow color whereas chlorine is more greenish. The color of each element in halogen group greatly vary from one another. This is because they can easily be reduced to –1 oxidation state by gaining an electron. Fluorine is the most reactive element among halogen elements due to its small size and lack of one electron to complete the octet rule. In nature, halogens are found as diatomic molecules. The electronegativity of halogens decreases down the group. Therefore, halogens can be found in ionic compounds as well as in covalent compounds. Since they lack one electron in order to fill their outermost orbital, they react with atoms or ions to obtain this electron. However, halogens are highly reactive because they all have electron configurations ending with ns 2np 5. For example, the chemical reactivity will decrease down the group due to the increase of the atomic size. There are patterns of varying the physical and chemical properties down the group of halogens in the periodic table. Iodine exists in the solid form while Astatine is a radioactive element.įigure 01: Halogens: chlorine gas, bromine liquid, and iodine solid (left to right) Fluorine and chlorine exist as gases bromine exists as a liquid. In the group of halogens, all three states of matter can be observed at standard temperature and pressure. They were given the name halogen because they all form sodium salts of similar properties such as sodium fluoride (NaF) and sodium chloride (NaCl). These 5 elements together are called halogens. This group includes fluorine (F), chlorine (Cl), bromine (Br), iodine (I) and Astatine (At). Halogen is a term used to represent the chemical elements in the group 7 of the periodic table of elements. 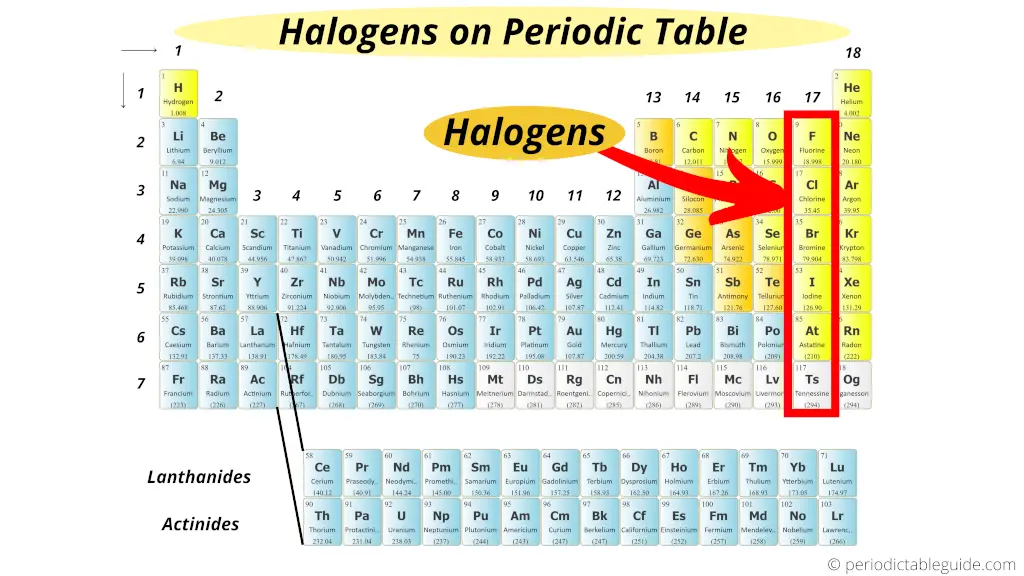
Key Terms: Electronegativity, Halogen, Inert, Noble Gas, Reactivity, Xenon What is Halogen What is the Difference Between Halogen and Xenon What are the Similarities Between Halogen and XenonĤ. – Definition, Properties, Reactions and their Usesģ. 
The main difference between halogens and xenon is that halogens have a single unpaired electron in their outermost orbital whereas xenon has no unpaired electrons in its orbitals. It is generally unreactive, but it can undergo several chemical reactions under extreme conditions. Xenon is a noble gas that is in the group 8 of the periodic table. They have varying physical and chemical properties. Halogens are the chemical elements in the group 7 of the periodic table and include F, Cl, Br, I and At. Halogens and Xenon are chemical elements that are included in group 7 and group 8 of the periodic table, respectively. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
